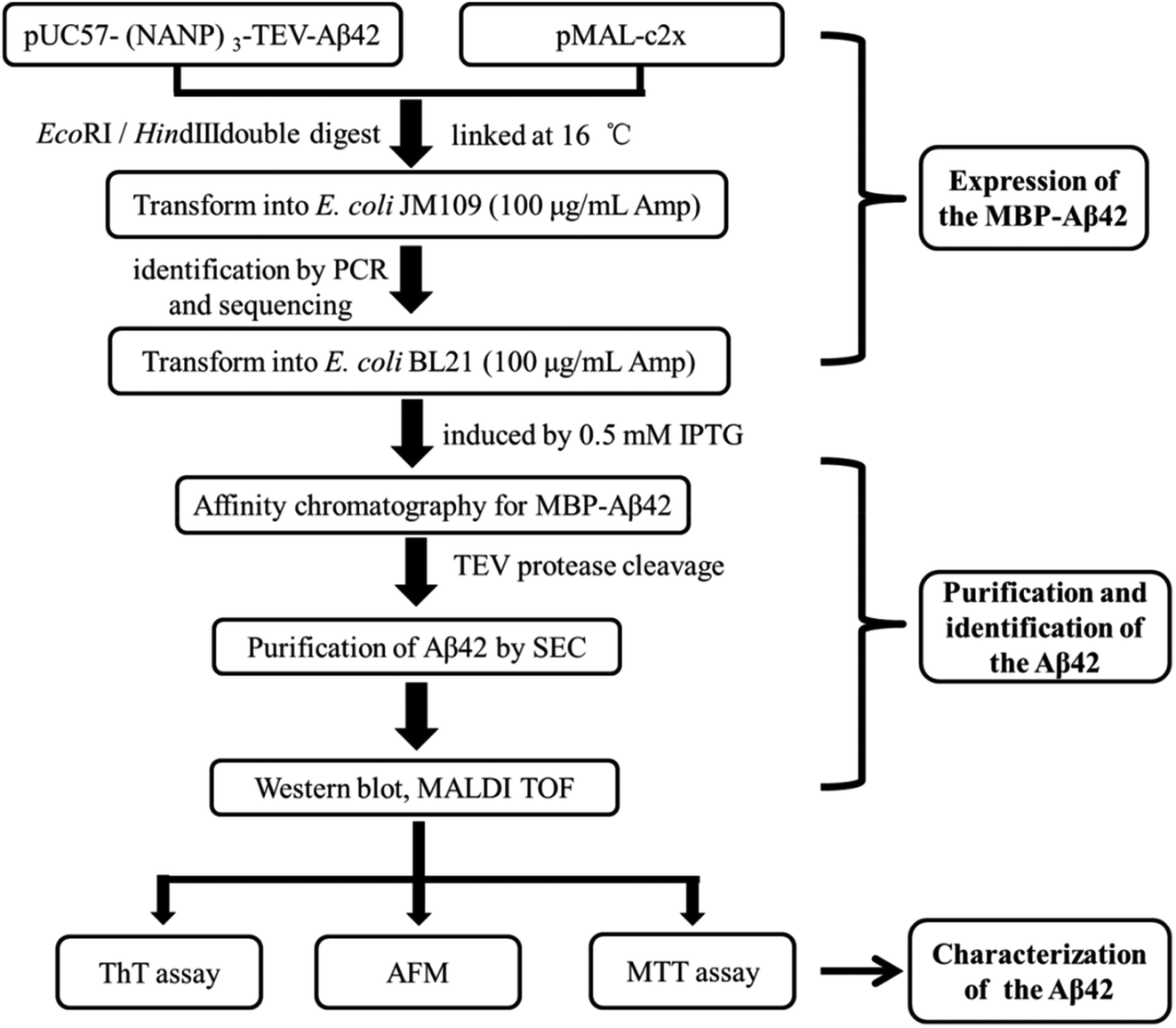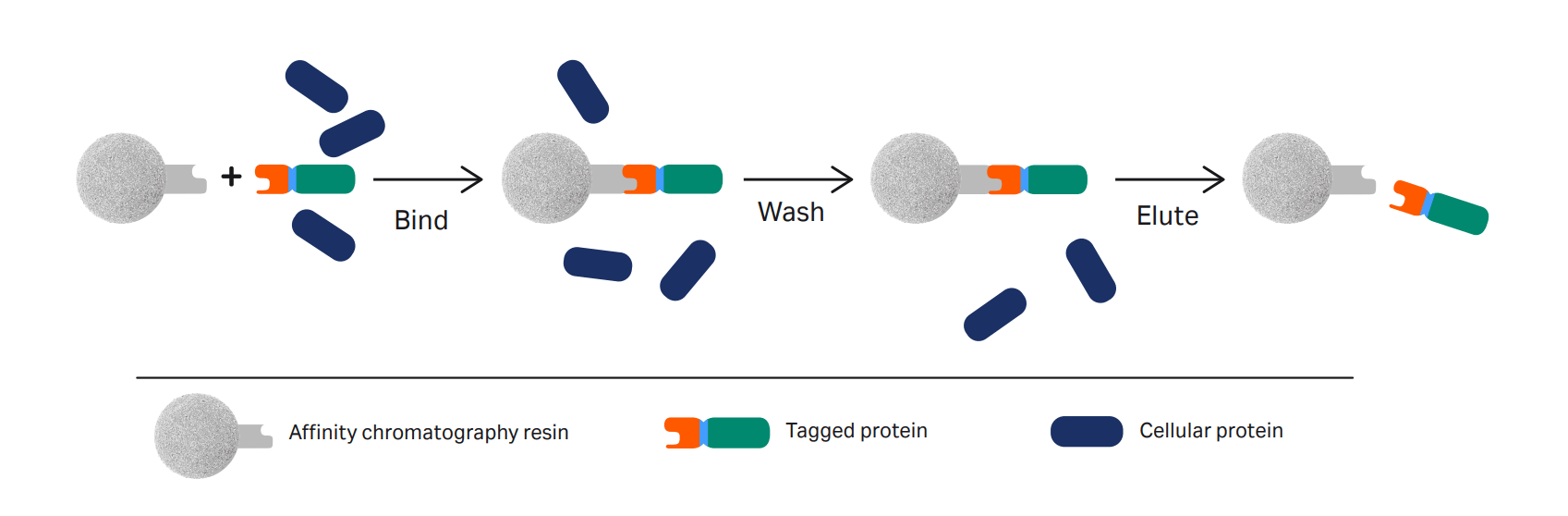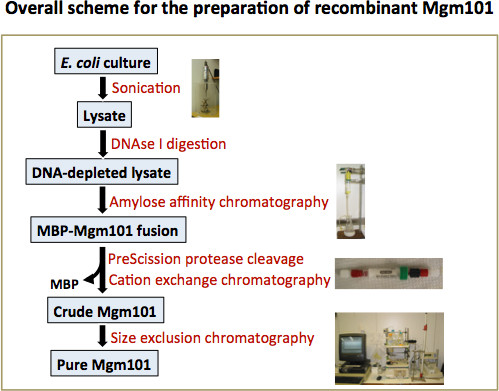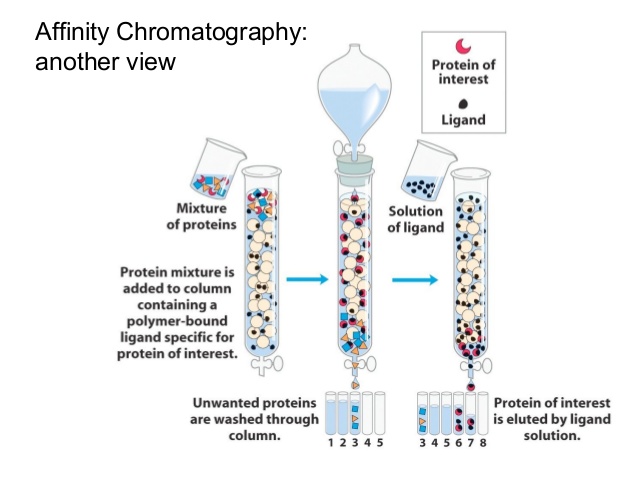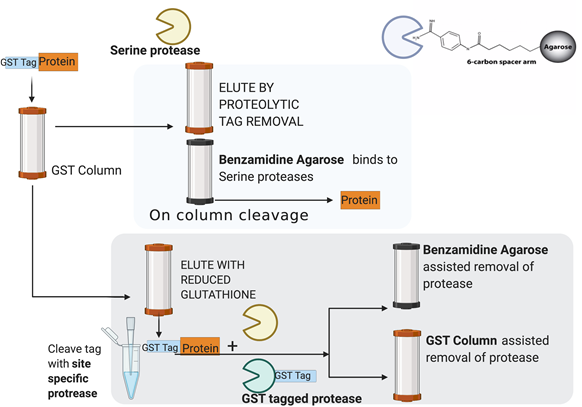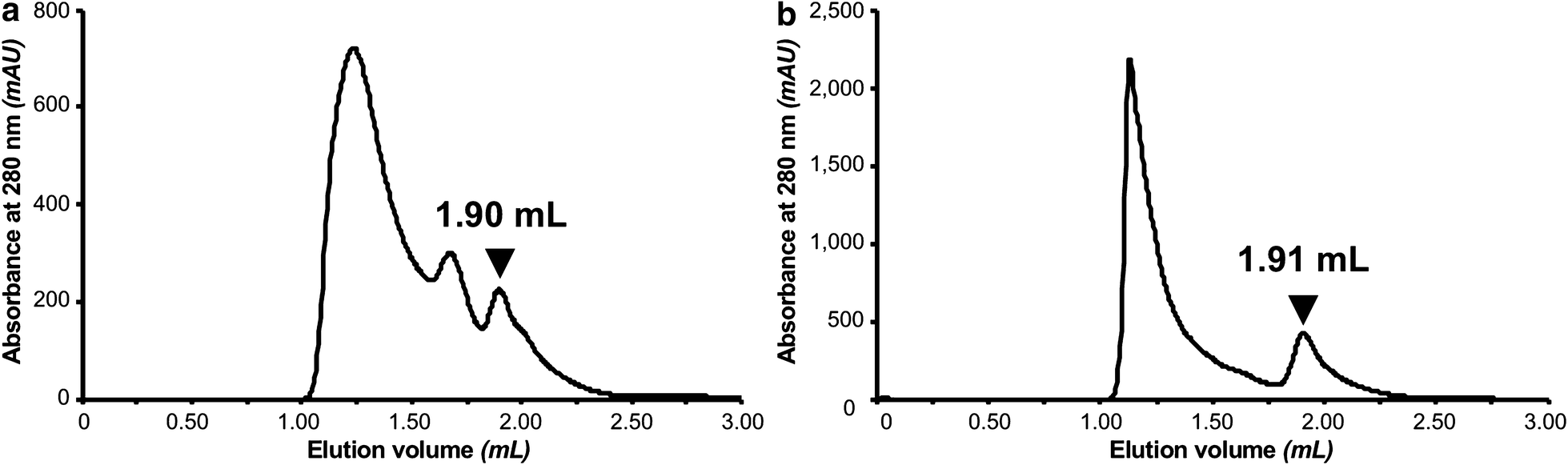
One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text
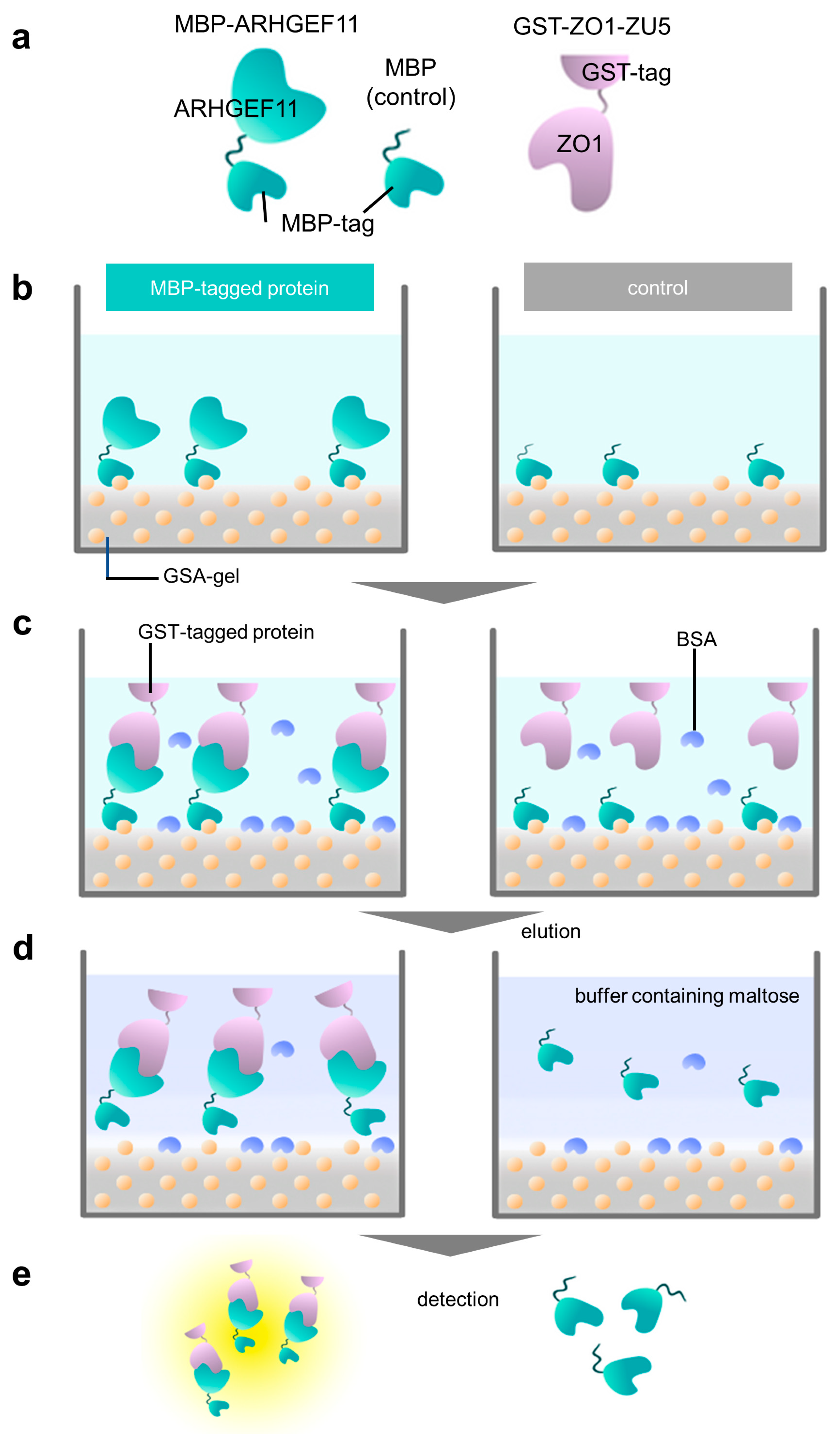
MPs | Free Full-Text | A Cost-Effective Immobilization Method for MBP Fusion Proteins on Microtiter Plates Using a Gelatinized Starch–Agarose Mixture and Its Application for Convenient Protein–Protein Interaction Analysis

Protein and antibody purification followed by immunoprecipitation of MYB and GATA zinc finger-type maize proteins with magnetic beads - ScienceDirect
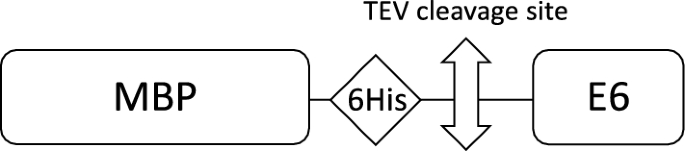
Strategies for bacterial expression of protein–peptide complexes: Application to solubilization of papillomavirus E6
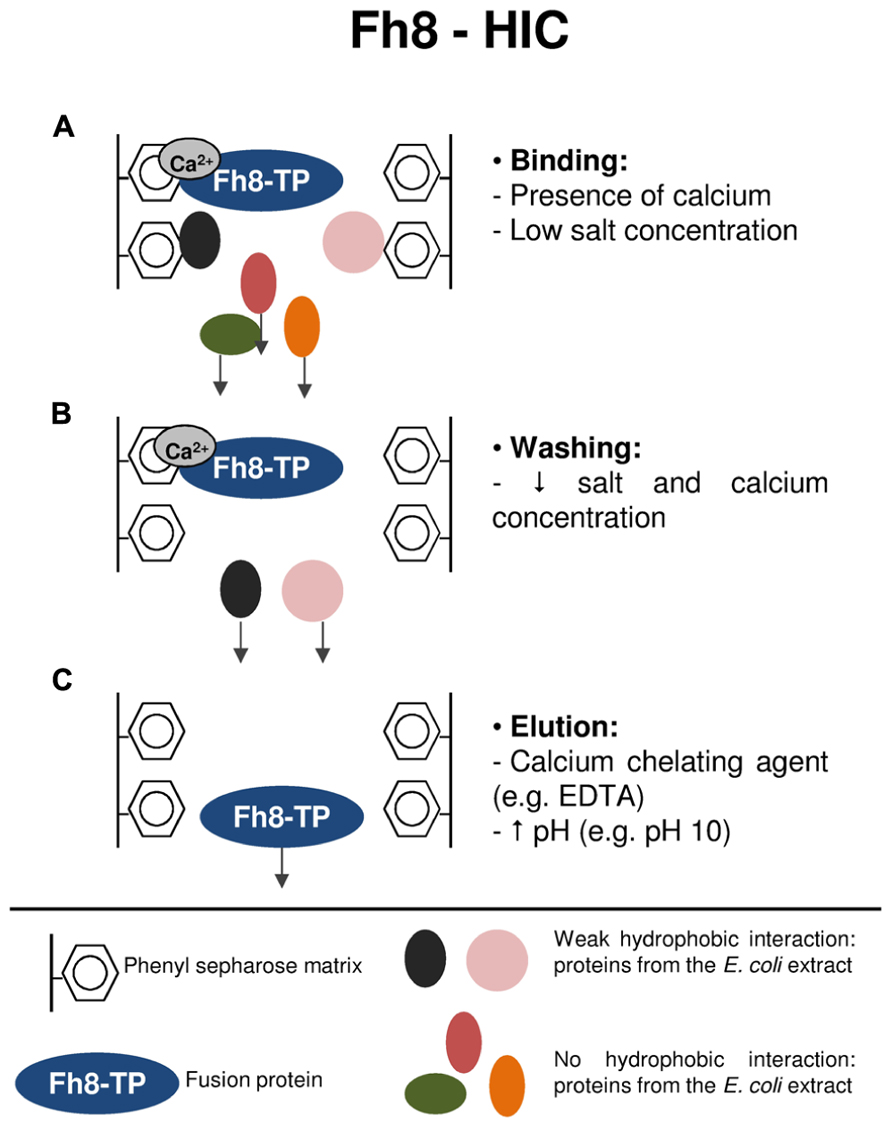
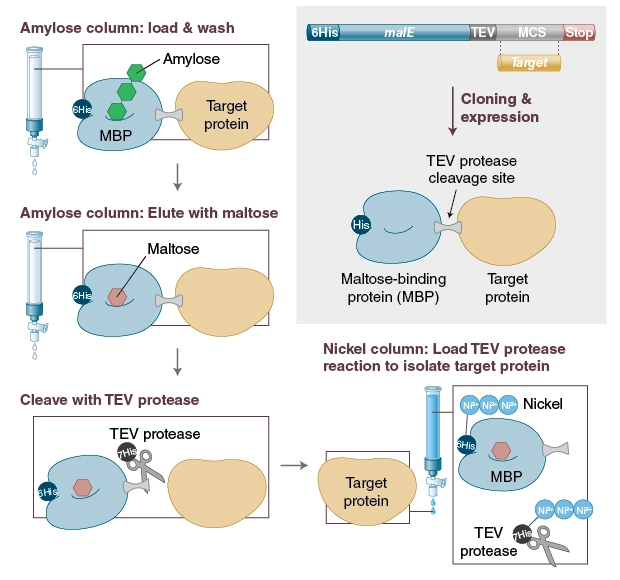
Frontiers | Fusion tags for protein solubility, purification and immunogenicity in Escherichia coli: the novel Fh8 system
One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text

Purification of hetero-oligomeric protein variants using a modified tandem affinity purification approach - ScienceDirect
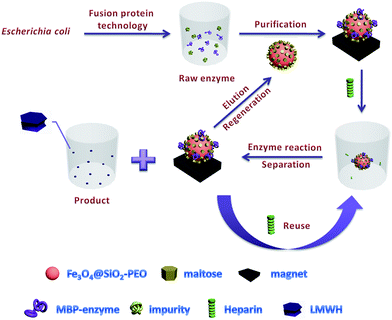
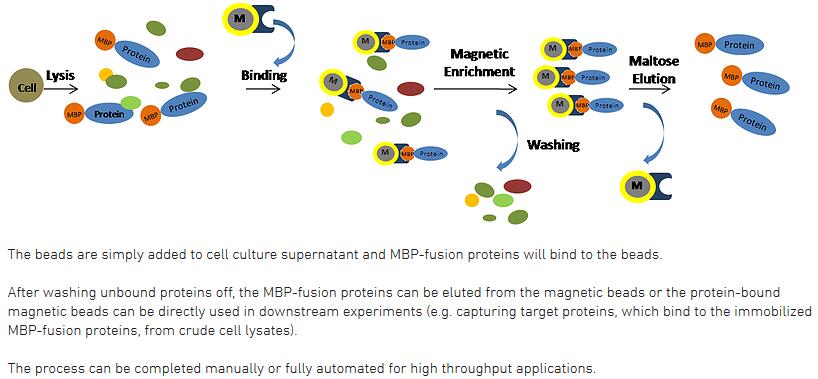
Magnetic nanoparticles for the affinity adsorption of maltose binding protein (MBP) fusion enzymes - Journal of Materials Chemistry (RSC Publishing) DOI:10.1039/C2JM16778F
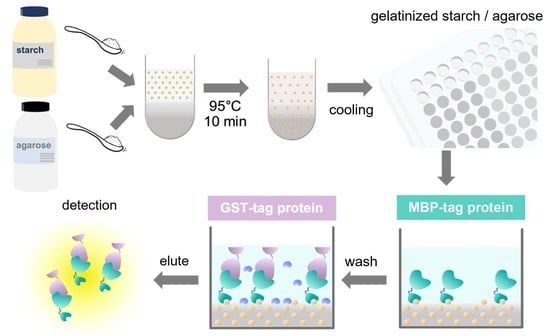
MPs | Free Full-Text | A Cost-Effective Immobilization Method for MBP Fusion Proteins on Microtiter Plates Using a Gelatinized Starch–Agarose Mixture and Its Application for Convenient Protein–Protein Interaction Analysis

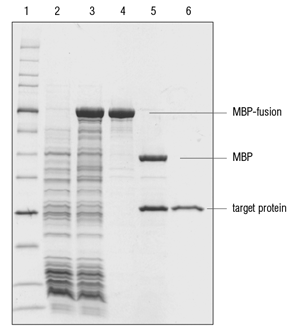
MS2BP-MBP fusion protein preparation. SDS-PAGE analysis showing the... | Download Scientific Diagram

Purification of hFGF21 from soluble MBP-hFGF21 expressed in E. coli... | Download Scientific Diagram
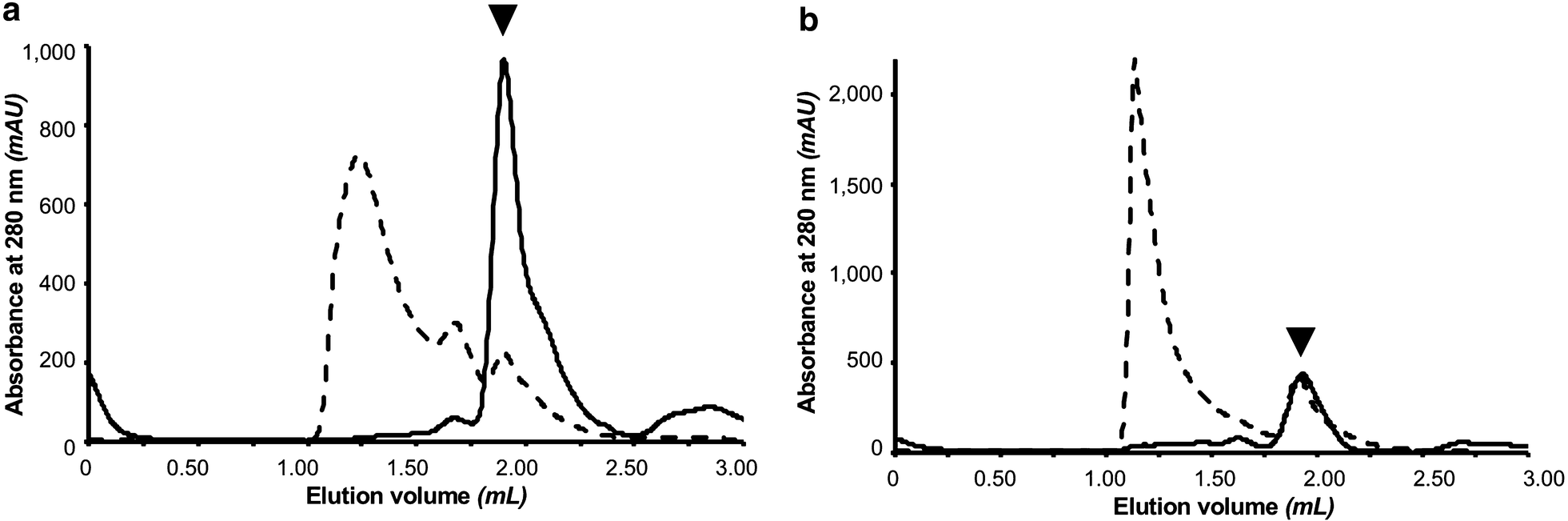
One-step affinity purification of fusion proteins with optimal monodispersity and biological activity: application to aggregation-prone HPV E6 proteins | Microbial Cell Factories | Full Text
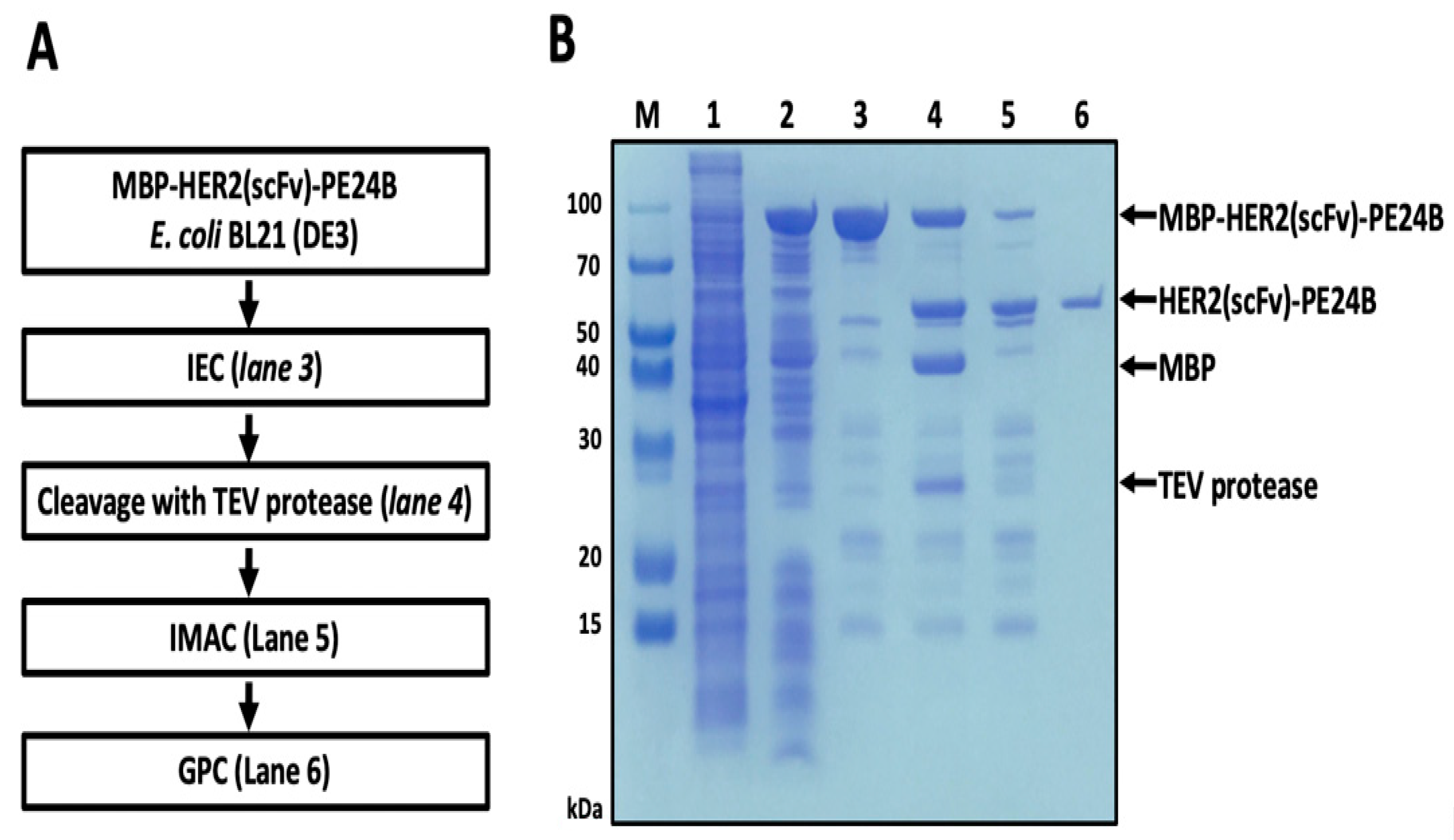
IJMS | Free Full-Text | Soluble Cytoplasmic Expression and Purification of Immunotoxin HER2(scFv)-PE24B as a Maltose Binding Protein Fusion