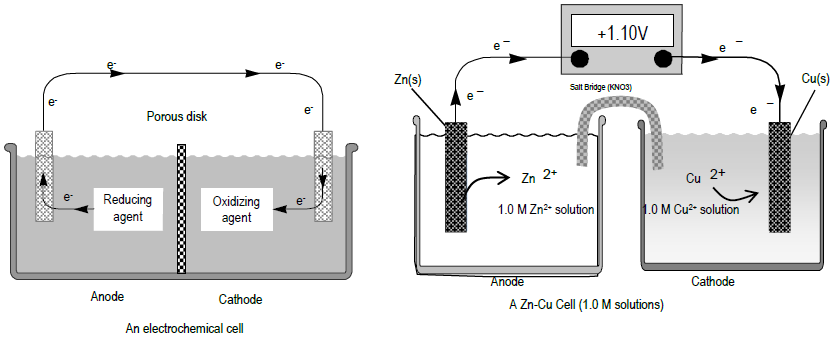
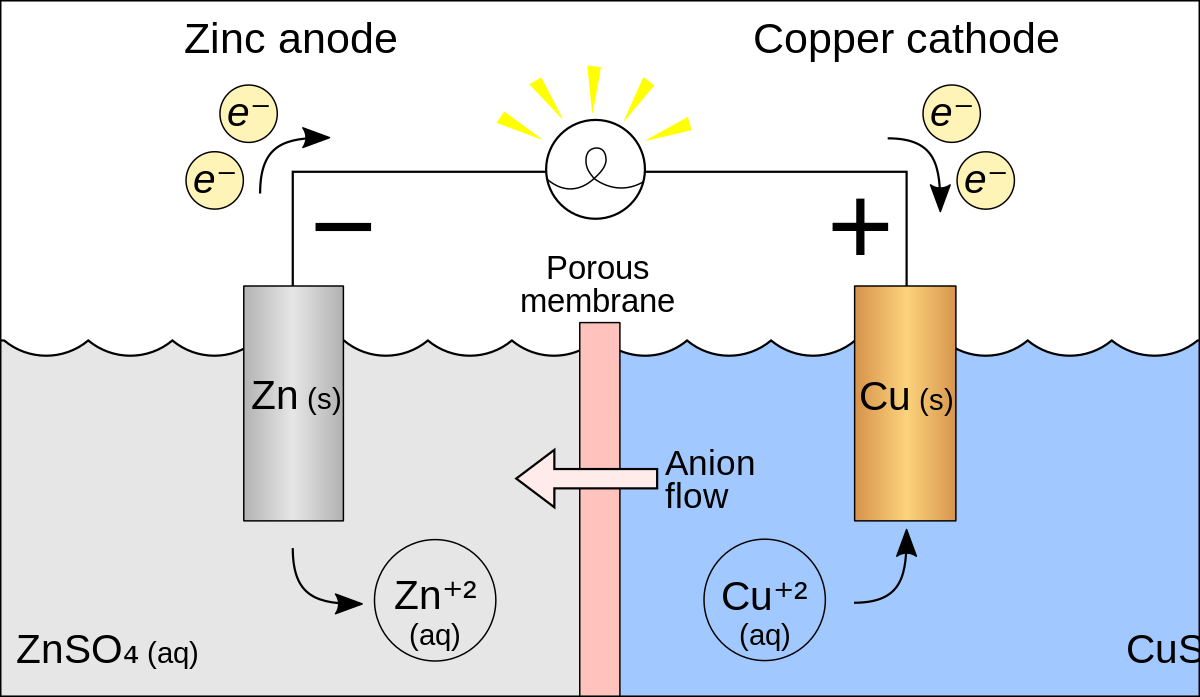
A voltaic cell is set up 25° C with the following half-cells: - Sarthaks eConnect | Largest Online Education Community

pls answer the whole question 14 A voltaic cell is set up at 25 C with the following - Chemistry - Electrochemistry - 13701485 | Meritnation.com

A voltaic cell in up 25oC with the following half cells:Al/Al3+(0.001M) and Ni/Ni2+(0.50M)Write an equation the reaction that occurs when the cell generates an electric current and determine the cell potential. ENi2+/Ni=−0.25V

✓ Solved: A silver concentration cell is set up at 25^°C as shown below: The AgCl(s) is in excess in...
A voltaic cell is set up at 25°C with the following half cells. Al | Al^3+ (0.0010 M) and Ni^2+ (0.50 M) | Ni - Sarthaks eConnect | Largest Online Education Community
A copper_ silver cell is set up. The copper ion concentration is 0.10 M. The concentration of silver ion is not know.the cell potential when measured was 0.422V determine the concentration of
A voltaic cell is setup at 25°C with the half cells Ag^+ (0.001 M) Ag and Cu^2+ (0.10 M) Cu. What should be its cell potential ? - Sarthaks eConnect | Largest Online Education Community





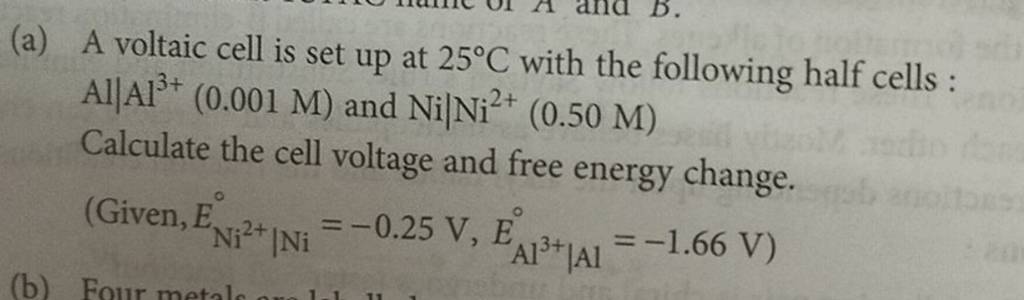
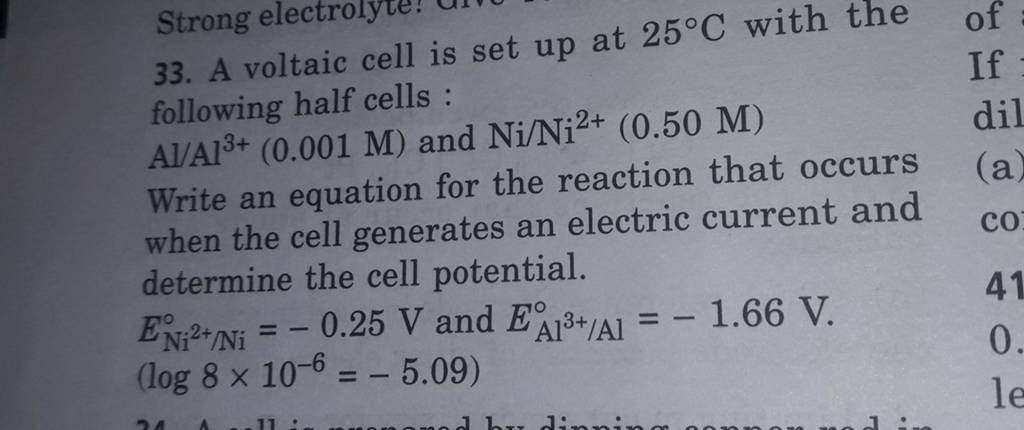


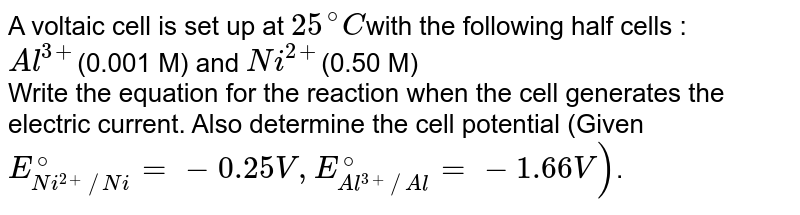
![Kannada] A voltic cell is set up at 25^@C with the half cells Ag^(+) Kannada] A voltic cell is set up at 25^@C with the half cells Ag^(+)](https://d10lpgp6xz60nq.cloudfront.net/ss/web-overlay-thumb/7681973.webp)